Hi Jenn,
I agree with Simon that having Review Type on the Study level screen
would be a great product enhancement, so I encourage you to submit
that idea. (Thanks!)
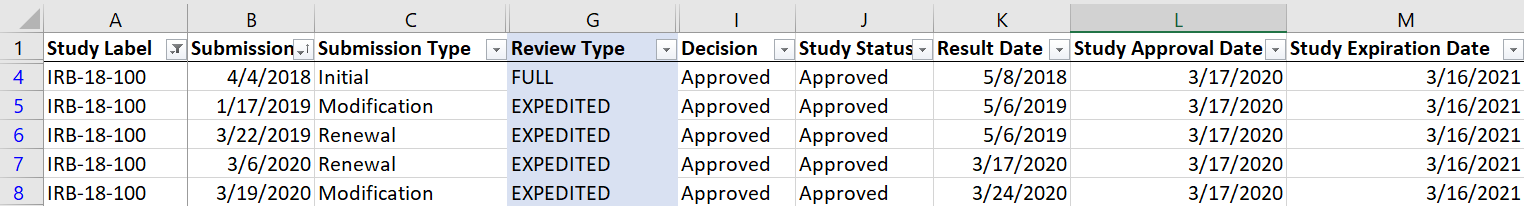
In the meantime, what I suggest is to create a report which includes Review
Type (if you haven't already done so) and sort by Study Label then Submission
Date (see pic below) to get a full picture of study history and current Review Type.
In this example, you can see that study IRB-18-100 had an Initial review type of Full,
but after a Modification, it was changed to Expedited and remains that way to date.
You could also decide to sort by Result Date rather than Submission Date.
I hope this helps!
Thanks,
Matty
------------------------------
Matty Gilreath
Customer Success Manager
Cayuse
matty.gilreath@cayuse.com------------------------------
Original Message:
Sent: 04-13-2021 08:24
From: Simon Helton
Subject: Cayuse Human Ethics - Tracking Study Level Determinations
Hi Jenn,
Sorry for the slow reply on this!
I asked around internally and didn't learn of any current workaround in this, but I hope you'll submit it as a product idea so that it can get formally considered. Thank you!
Simon
------------------------------
Simon Helton
He/him
Community Manager
Cayuse
Original Message:
Sent: 03-29-2021 20:56
From: Jennifer Dier
Subject: Cayuse Human Ethics - Tracking Study Level Determinations
Hi All,
A question has come up as my institution moves towards our Cayuse Human Ethics "go live" date. We're wondering if other institutions have a way (work-around) to track a study's current overall Review Type (exempt/limited/review/expedited/full board) at the Study Details level. Since Review Type is noted in the Submission tab for each submission, and a submission's Review Type might be different that that of the overall study, I'm wondering how others are handling this. For example, a study for which the initial submission was given a Review Type of full board (which we would consider to be the review type of the overall study) but a later modification submission was given the Review Type of expedited. Is this a non-issue, since the Review Type can be found in the Submission tab? I thank you in advance for your insight.
Best,
Jenn
------------------------------
Jennifer Dier
Research Compliance Analyst
University of California, Santa Cruz
jdier@ucsc.edu
------------------------------